Q.21 For product formation from only one type of reactant (e.g. A → product), the CORRECT match for the order of the reaction (given in Column I) with the half-life […]
Blog
Hybridisation of Central Atom in I₃⁻, PCl₃, BF₃ and CO₂
Q. 20 A few species are given in Column I. Column II contains the hybrid orbitals used by the central atom of the species for bonding. The CORRECT match for […]
Valine Isoelectric Point Calculation
Q.19 Consider the two 𝑝𝐾𝑎values of valine as 2.32 and 9.62. The isoelectric point (pI)of this amino acid is ______. (rounded off to two decimal places) The isoelectric point (pI) […]
[Ni(CN)4]2− [Ni(CO)4] [NiCl4]2− Geometry Magnetism
Q.18 The CORRECT statement(s) about [Ni(CN)4]2−, [Ni(CO)4] and [NiCl4]2− is(are)(Given: Atomic number of Ni: 28)(A) Both [Ni(CN)4]2− and [Ni(CO)4] are square planar complexes.(B) [Ni(CN)4]2− is diamagnetic and [NiCl4]2− is paramagnetic.(C) […]
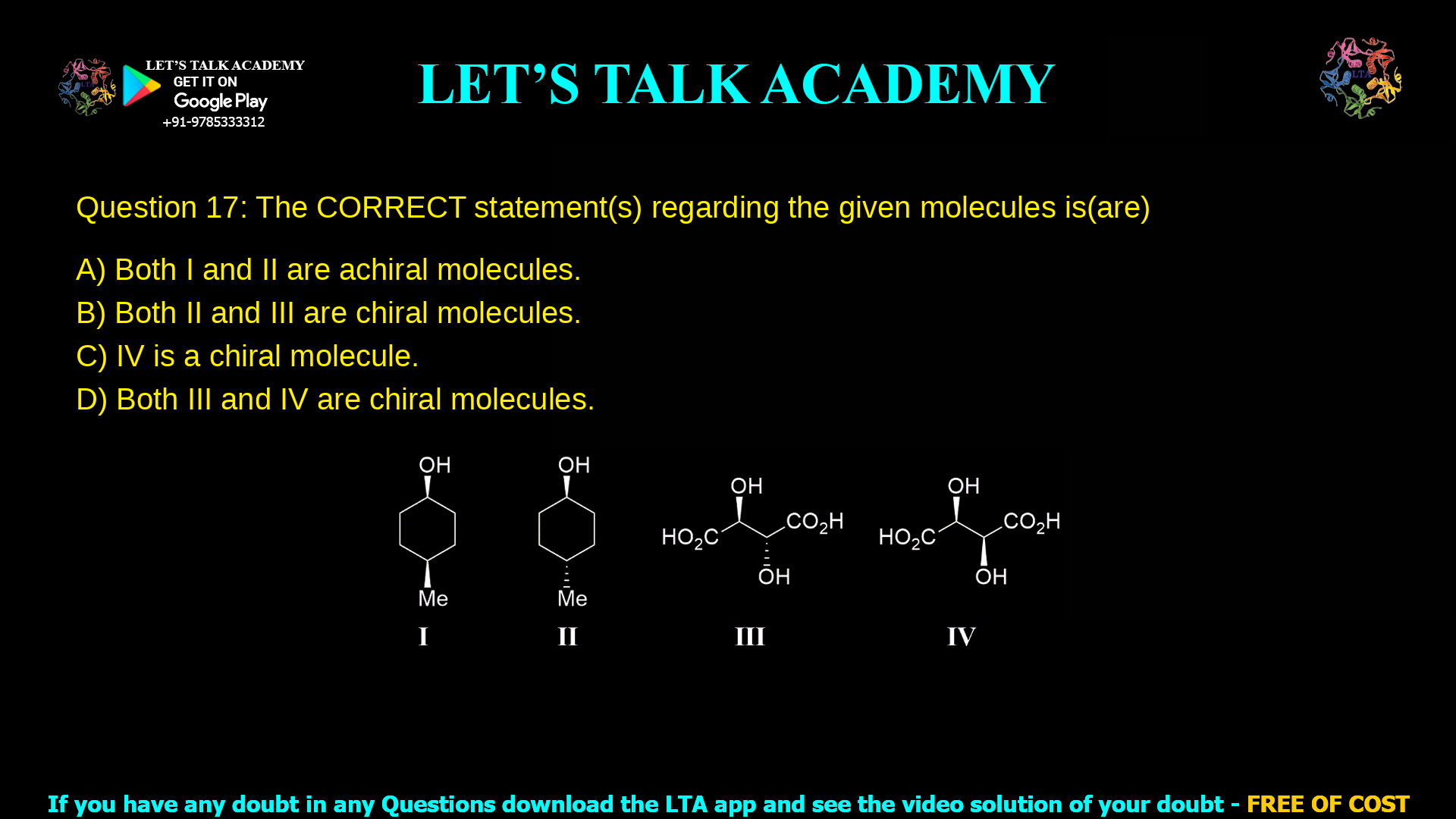
Chirality of Cyclohexane and Tartaric Acid Isomers
Q. 17 The CORRECT statement(s) regarding the given molecules is(are)(A) Both I and II are achiral molecules.(B) Both II and III are chiral molecules.(C) IV is a chiral molecule.(D) Both […]
Isothermal Expansion of Ideal Gas
Q. 16 The isothermal expansion of one mole of an ideal gas from 𝑉𝑖 to 𝑉𝑓 at temperature,T occurs in two ways.Path I: a reversible isothermal expansion;Path II: free expansion […]
Arrhenius Equation Plot
Q. 15 The temperature dependence of reaction rates is generally given by the Arrheniusequation. A plot of ln 𝑘𝑟 against 1/𝑇 is a straight line from which the pre–exponential factor […]
Standard Enthalpy of Formation H2O(g)
Q. 14 The standard enthalpy of the reaction,C (graphite) + H2O (g) → CO (g) + H2 (g) is found to be +131.3 kJ mol–1and the ∆𝑓𝐻0 value for CO […]
Bond Order of N22−
Q. 13 The bond order in 𝑁22− species is(A) 2(B) 2.5(C) 3(D) 3.5 The bond order of N22− is 2, so the correct option is (A) 2. Introduction The bond […]
Correct Order of Boiling Points for Hydrogen Halides
Q. 12 The CORRECT order of boiling points for the hydrogen halides is(A) HF > HI > HBr > HCl(B) HF > HCl > HBr > HI(C) HI > HBr […]
![Q.21 For product formation from only one type of reactant (e.g. A → product), the CORRECT match for the order of the reaction (given in Column I) with the half-life expression (given in Column II) is ([A]0 is the initial concentration and kr is the rate constant) Column I Order Column II Half-life expression i. Zero P. ln 2 / kr ii. First Q. [A]0 / 2kr iii. Second R. 1 / (kr[A]0) S. 2kr / [A]0 (A) i–R, ii–P, iii–S (B) i–Q, ii–P, iii–R (C) i–S, ii–R, iii–Q (D) i−Q, ii−P, iii−S](https://www.letstalkacademy.com/wp-content/uploads/2025/12/slide_21-4.png)


![Q.18 The CORRECT statement(s) about [Ni(CN)4]2−, [Ni(CO)4] and [NiCl4]2− is(are) (Given: Atomic number of Ni: 28) (A) Both [Ni(CN)4]2− and [Ni(CO)4] are square planar complexes. (B) [Ni(CN)4]2− is diamagnetic and [NiCl4]2− is paramagnetic. (C) Both [Ni(CO)4] and [NiCl4]2− are paramagnetic. (D) [Ni(CN)4]2− is square planar and [NiCl4]2− is tetrahedral in shape.](https://www.letstalkacademy.com/wp-content/uploads/2025/12/slide_18-4.png)