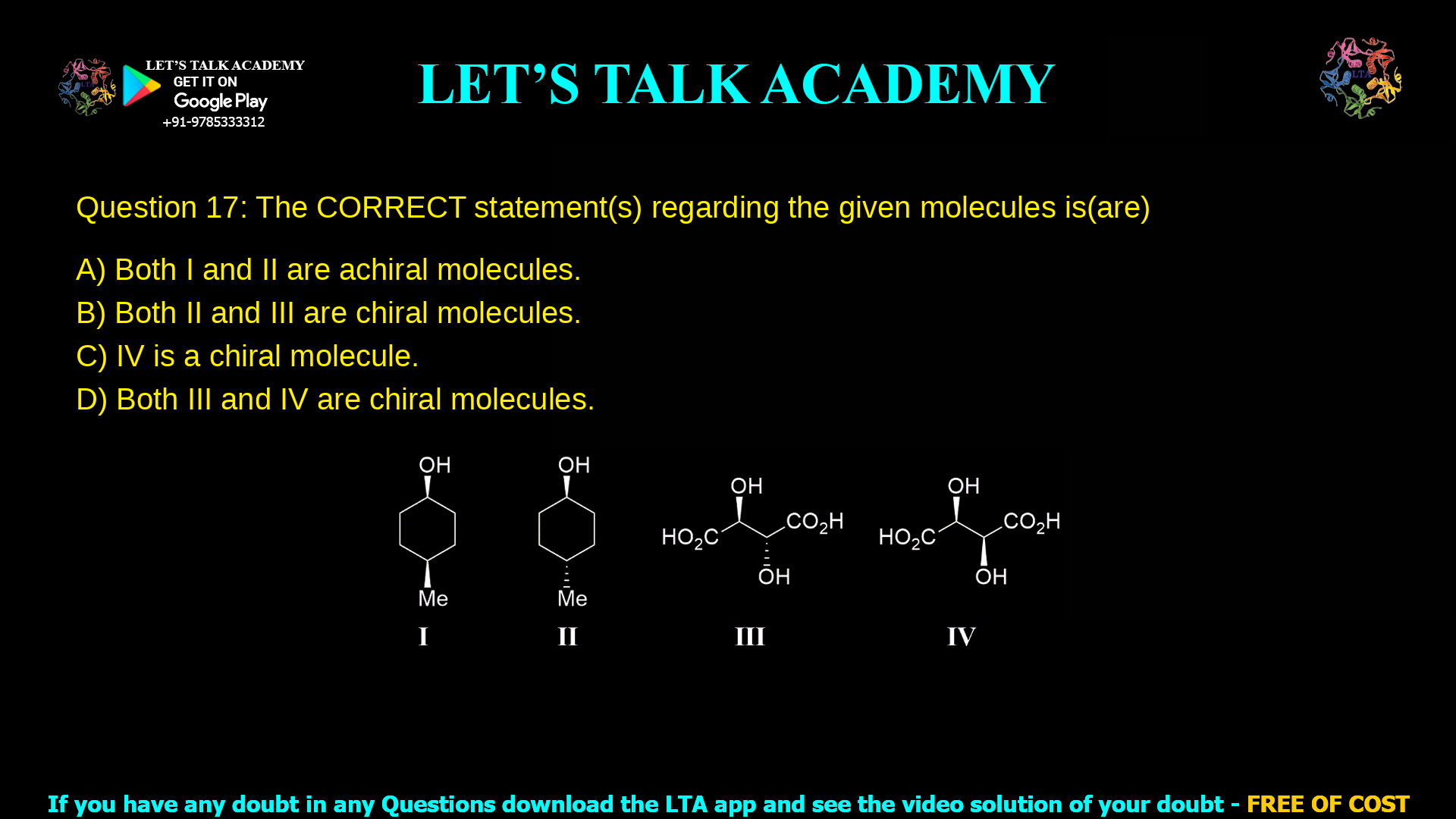
Q. 17 The CORRECT statement(s) regarding the given molecules is(are)
(A) Both I and II are achiral molecules.
(B) Both II and III are chiral molecules.
(C) IV is a chiral molecule.
(D) Both III and IV are chiral molecules.
The correct answer is that molecules II and III are chiral, while I and IV are achiral; therefore option (B) is correct.
Introduction
In CSIR NET and GATE stereochemistry, questions on chirality often combine substituted cyclohexanes and tartaric acid–type systems, testing understanding of symmetry and stereocenters.
This question asks which of the four given molecules (I–IV) are chiral or achiral, so the key is to analyse mirror planes and meso behaviour in each structure.
Step‑wise analysis of molecules I–IV
Molecule I
-
Structure I is a substituted cyclohexane with an OH and a Me group on adjacent carbons; in the standard drawing for this question, both substituents are on the same side of the ring (cis‑1,2‑substitution) giving an internal mirror plane through the ring and bisecting the C–C bond between them.
-
Because there is a plane of symmetry, the molecule is superimposable on its mirror image and hence achiral despite the apparent stereocenters.
Molecule II
-
Structure II is the corresponding cyclohexane where OH and Me on adjacent carbons are on opposite sides of the ring (trans‑1,2‑substitution), which removes the internal mirror plane present in I.
-
In the most stable chair conformation there is no plane or center of symmetry, so the configuration is non‑superimposable on its mirror image, making molecule II chiral.
Molecule III
-
Structure III is HO₂C–CH(OH)–CH(OH)–CO₂H where the two OH groups are on opposite sides (one wedge, one dash) giving the general tartaric acid formula with RR/SS type relationship between the two stereocenters.
-
This arrangement lacks an internal mirror plane and corresponds to optically active D‑ or L‑tartaric acid, so molecule III is chiral.
Molecule IV
-
Structure IV is the same HO₂C–CH(OH)–CH(OH)–CO₂H skeleton, but both OH groups are on the same side (both wedges or both dashes), giving a configuration where the two stereocenters are R and S (or S and R).
-
In this case there is an internal plane of symmetry passing vertically through the central C–C bond, so the compound is the meso‑tartaric acid, which is optically inactive and therefore achiral even though it has two stereocenters.
Evaluation of each option
Option (A): Both I and II are achiral molecules
-
Molecule I is achiral due to a mirror plane, but molecule II lacks any symmetry element that would make it superimposable on its mirror image and is chiral.
-
Hence option (A) is incorrect because it misclassifies molecule II.
Option (B): Both II and III are chiral molecules
-
As discussed, trans‑1,2‑substituted cyclohexane II has no plane of symmetry and is chiral, and tartaric acid isomer III corresponds to the optically active D/L form, which is also chiral.
-
Therefore option (B) is correct.
Option (C): IV is a chiral molecule
-
Molecule IV is the meso form of tartaric acid with an internal plane of symmetry, making it achiral despite having two stereocenters.
-
Thus option (C) is incorrect.
Option (D): Both III and IV are chiral molecules
-
While III is chiral, IV is a meso compound and achiral, so pairing them as both chiral is wrong.
-
Hence option (D) is incorrect.