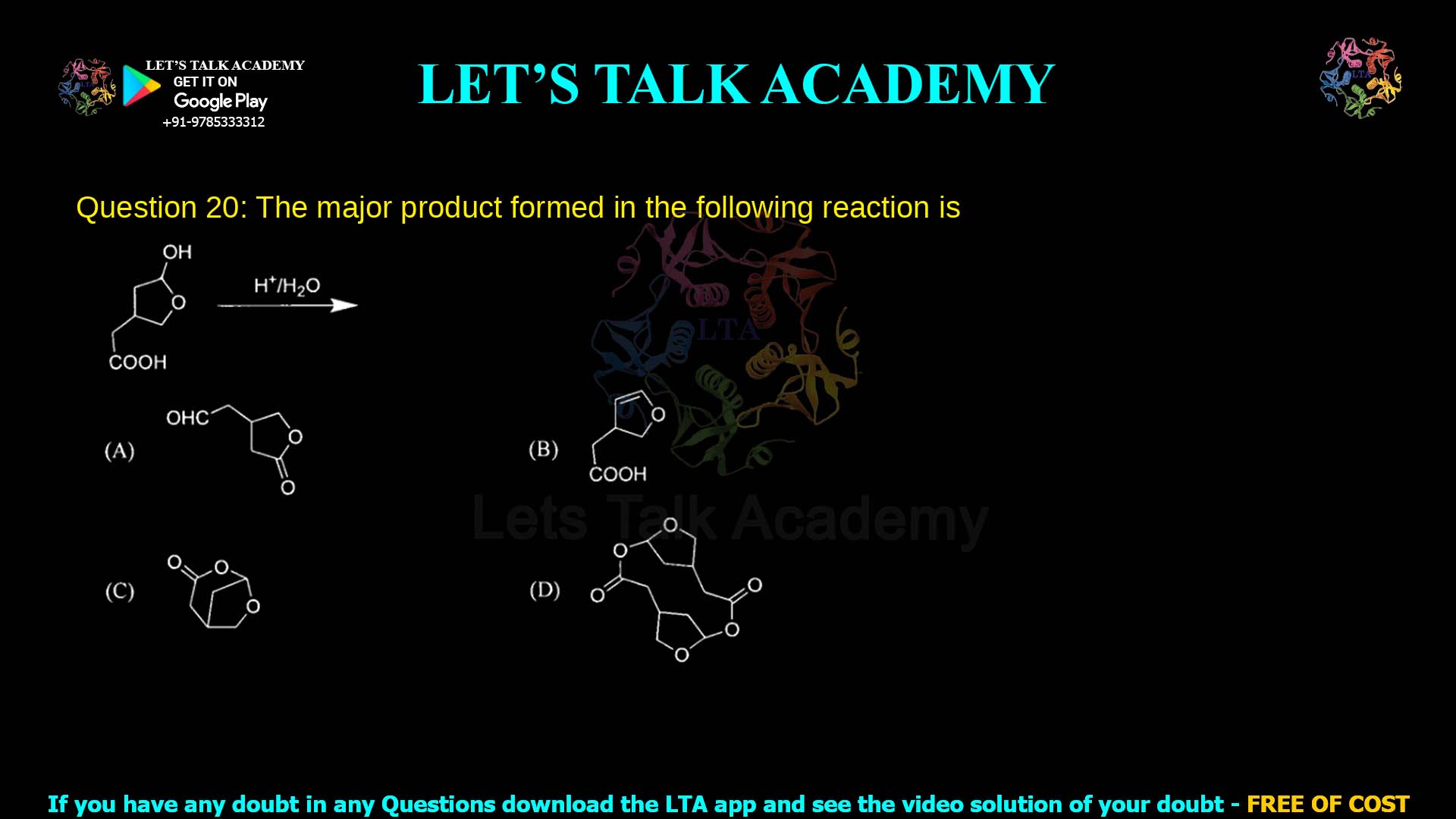
Q20. The major product formed in the following reaction is
The major product is the five‑membered lactone (option C) formed by intramolecular esterification of the γ‑hydroxycarboxylic acid under acid catalysis.
Introduction
In this article, the major product formed in the acid‑catalyzed reaction of a γ‑hydroxycarboxylic acid carrying a five‑membered backbone is analyzed to identify which lactone (or related structure) appears as the correct option in a typical CSIR‑NET multiple‑choice question. The discussion uses fundamental concepts of intramolecular esterification, ring size preference, and stability of γ‑lactones to arrive at the right answer and to eliminate the incorrect options.
Step‑by‑step solution
Nature of the starting compound
-
The given structure is a γ‑hydroxycarboxylic acid: three carbon atoms separate the hydroxyl group and the carboxylic acid group on a five‑membered ring framework.
-
Under acidic conditions, such γ‑hydroxy acids readily undergo intramolecular esterification to form γ‑lactones (five‑membered cyclic esters), which are thermodynamically very stable.
Mechanism under H⁺/H₂O
-
Protonation of the carbonyl oxygen of the carboxylic acid increases electrophilicity of the carbonyl carbon.
-
The intramolecular hydroxyl group attacks this activated carbonyl, giving a tetrahedral intermediate.
-
Proton transfer followed by loss of water gives an ester linkage within the same molecule, closing a five‑membered ring – a γ‑lactone.
-
Five‑ and six‑membered rings form preferentially because they have minimal ring strain, so the five‑membered lactone is the favored product for a γ‑hydroxy acid.
Thus, the major product is a five‑membered cyclic ester (γ‑lactone), represented by option C.
Option‑wise analysis
Option A
-
Option A shows a ring bearing an aldehyde (–CHO) plus an internal ester function.
-
Conversion of a γ‑hydroxycarboxylic acid to an aldehyde would require reduction or oxidative cleavage steps, which are not available in simple acid‑catalyzed aqueous conditions.
-
Therefore this structure is not consistent with the intramolecular esterification pathway, so option A is incorrect.
Option B
-
Option B retains a free carboxylic acid group but introduces an additional small cyclic acetal or heterocycle separate from the COOH.
-
Under H⁺/H₂O with this substrate, the more favorable transformation is cyclization between the existing OH and COOH to form a lactone, not preservation of COOH with a different ring system.
-
Hence option B does not represent the typical product of γ‑hydroxy acid cyclization and is incorrect.
Option C
-
Option C is a five‑membered lactone in which the original hydroxyl and carboxyl groups have combined to form a cyclic ester, eliminating water.
-
This matches the known behavior of γ‑hydroxycarboxylic acids, which spontaneously cyclize to γ‑lactones under acidic conditions because such rings are especially stable.
-
Therefore option C is the correct major product.
Option D
-
Option D contains a dimeric structure with two lactone units fused through acetal‑like linkages.
-
Dimerization would require intermolecular reaction between two molecules, which is disfavored relative to the fast intramolecular cyclization that gives a single γ‑lactone; intramolecular processes are entropically and kinetically preferred.
-
As such, option D does not represent the major product and is incorrect.
Key takeaway
Under H⁺/H₂O, a γ‑hydroxycarboxylic acid on a five‑membered backbone undergoes intramolecular esterification to give a γ‑lactone, making option C the correct answer in this MCQ.