11. Which of the following statements are true? a. Vmax of an enzyme remains constant with increasing enzyme concentrations. b. KM of an enzyme for a substrate remains constant with […]
Blog
Octane Combustion CO2 Calculation
10. If 1.14 Kg of octane are burned in the complete combustion reaction shown below, how many Kg of carbon dioxide will be produced (atomic mass of C, O and […]
How to Find Chiral Centres in Complex Organic Molecules
9. How many chiral centres does this molecule below have: a. 2 b. 7 c. 8 d. 9 Introduction Exam questions like “How many chiral centres does this molecule below have?” frequently […]
Hydropathy Plot of a Protein
8. In the below hydropathy plot of a protein, positive values are regions of the protein that are considered hydrophobic based on primary amino acid sequence. What type of protein […]
pH of Neutral Water at 65°C
7. At 25 °C and zero ionic strength, water self-dissociation results in: Kw = [H3O+][OH-] = 1.0×10−14 If it is known that Kw increases with increasing temperature, what will be […]
H2 + F2 → 2HF Bond Energy Enthalpy Calculation
6. Using the bond energy data given in the parenthesis, what would be the expected enthalpy for the following reaction H2(g) + F2 (g) à 2HF (g) (H-H: 432 kJ/mol; […]
NaHCO3 + HCl pH Calculation
5. 8.4 mg NaHCO3 powder was slowly added to 10 ml 0.03 M HCl in a 50ml beaker with stirring. After all effervescence has seized, what should be the pH […]
CH3CHO and C6H5CH2CHO Distinction
4. CH3CHO and C6H5CH2CHO can be distinguished chemically by: a. Tollen’s reagent test b. Fehling solution test c. Benedict test d. Iodoform test CH3CHO (acetaldehyde) and C6H5CH2CHO (phenylacetaldehyde) can be […]
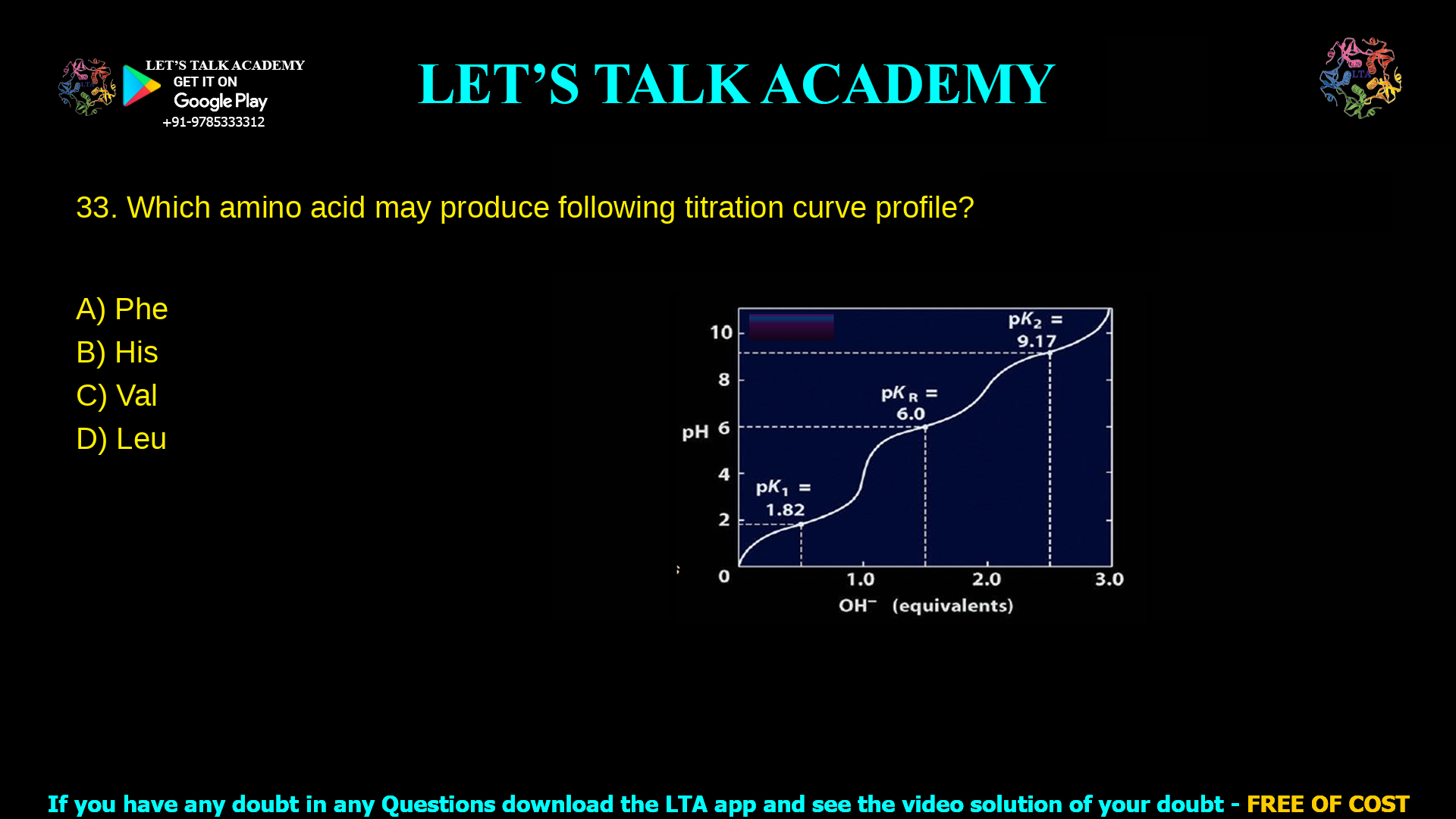
Detailed Explanation of Histidine Titration Curve and Amino Acid Options
3. Which amino acid may produce following titration curve profile? a. Phe b. His c. Val d. Leu Histidine is the amino acid that produces the given titration curve with […]
pKa 4.2: How to Find the pH at Which 95% of a Monobasic Organic Acid Is Ionized
2. The pKa of a monobasic organic acid is 4.2. The pH at which 95% of the acid will be in ionized form is: a. 5.02 b. 5.38 c. 2.92 […]




![7. At 25 °C and zero ionic strength, water self-dissociation results in: Kw = [H3O+][OH-] = 1.0×10−14 If it is known that Kw increases with increasing temperature, what will be the pH of neutral water at 65°C? a. Slightly less than 14 b. Equal to 7 c. Slightly less than 7 d. Slightly more than 7](https://www.letstalkacademy.com/wp-content/uploads/2025/12/slide_37-2.png)