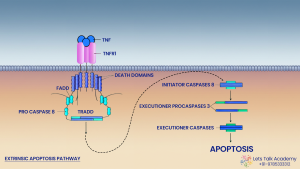
Extrinsic pathway of Apoptosis
The extrinsic pathway of apoptosis is a type of programmed cell death that is initiated by external signals. It begins when specific ligands (such as FasL or TNF-α) bind to death receptors (like Fas or TNFR1) on the cell surface. This triggers the formation of a death-inducing signaling complex (DISC), which activates caspase-8, leading to a cascade of caspase activation and ultimately cell death. This pathway plays a key role in immune system regulation and the elimination of harmful or infected cells.
Types of Death Receptor in Extrinsic pathway of Apoptosis
| Death Receptor | Ligand | Also Known As |
|---|---|---|
| Fas (CD95/APO-1) | Fas Ligand (FasL/CD95L) | Fas receptor |
| TNFR1 (CD120a) | TNF-α | Tumor necrosis factor receptor 1 |
| DR4 (TRAIL-R1) | TRAIL | Death receptor 4 |
| DR5 (TRAIL-R2) | TRAIL | Death receptor 5 |
Tumor Necrosis Factor Receptor (TNFRs)
Tumor Necrosis Factor Receptors (TNFRs) are a group of cell surface receptors that bind Tumor Necrosis Factor (TNF) ligands, playing a critical role in inflammation, immune regulation, and apoptosis. TNFRs are typically type one transmembrane protein which is having 2 to 4 identical cystine rich domain in extracellular portion. The TNFR 1 induces the formation of a secondary, cytoplasmic signalling complex that contains Receptor Interacting Protein Kinase 1 (RIP K1) together with FADD and caspase-8, referred as ripoptosis.
Key Features of Tumor Necrosis Factor Receptors (TNFRs):
-
Types: The main members include TNFR1 (p55/CD120a) and TNFR2 (p75/CD120b).
-
Ligands: Their main ligand is TNF-α, a pro-inflammatory cytokine.
-
TNFR1: Widely expressed, contains a death domain, and can trigger apoptosis via the extrinsic pathway.
-
TNFR2: Mainly expressed in immune cells; promotes cell survival and immune activation rather than apoptosis.
Extrinsic Apoptotic Signalling via TNFR1:
When TNF-α binds to TNFR1, a series of signalling events occur, which can lead to apoptosis under certain conditions:
1. Ligand Binding:
-
TNF-α (trimeric) binds to the extracellular domain of TNFR1 (Receptor is also Trimer).
-
This exposes its death domain of TNFR1 in the cytoplasm.
-
It recruits TRADD, which also has a DD, via DD–DD interaction.
2. Adaptor Recruitment:
-
TRADD (TNF Receptor-Associated Death Domain) binds to the TNFR1 death domain. TRADD also has Death Effector Domain (DED).
3. DISC Formation:
-
TRADD has both a DD and a DED.
-
DD interacts with death receptors.
-
DED interacts with procaspase-8 (which also has a DED).
-
-
TRADD recruits pro-caspase-8 to form the Death-Inducing Signalling Complex (DISC).
-
Within the DISC,DED and DD bind with the procaspase 8 and converted procaspases to caspase-8 pro-caspase-8 is cleaved and activated.
4. Caspase Cascade:
-
Active caspase-8 cleaves and activates executioner caspases like caspase-3, caspase-6, and caspase-7. Activated caspase-3 leads for the cytoskeleton degradation and also then it activates the CAD from the ICAD, now the CAD lead for the DNA fragmentation.
-
This leads to the systematic breakdown of cellular components and apoptosis.
Fas (CD95/APO-1) Signaling Pathway in the Extrinsic Apoptosis Pathway
The Fas receptor (CD95/APO-1) is a classic death receptor that triggers apoptosis upon binding its ligand, FasL (Fas Ligand/CD95L). This interaction plays a vital role in immune regulation and the elimination of harmful or unnecessary cells.
Step-by-Step Signalling Mechanism:
1. Ligand Binding and Receptor Trimerization
-
FasL (trimeric) binds to Fas receptor (CD95) on the target cell.
2. Death Domain Exposure and FADD Recruitment
-
The intracellular Death Domain (DD) of Fas becomes exposed.
-
FADD (Fas-Associated Death Domain) adaptor protein binds to Fas via DD–DD interaction.
3. DISC Formation (Death-Inducing Signaling Complex)
-
FADD has a second domain, the Death Effector Domain (DED).
-
Procaspase-8 or procaspase-10 (which also contain DEDs) bind to FADD through DED–DED interaction.
-
This results in the assembly of the DISC.
4. Caspase-8 Activation
-
Within the DISC, procaspase-8 is cleaved into its active form (caspase-8).
-
Active caspase-8 is released into the cytosol.
5. Downstream Caspase Cascade
-
Caspase-8 activates effector caspases like caspase-3, caspase-6, and caspase-7.
-
These caspases cleave cellular substrates, leading to:
-
DNA fragmentation
-
Membrane blebbing
-
Cell shrinkage
-
Apoptosis
-
Regulation of the Extrinsic Pathway of Apoptosis
The extrinsic pathway of apoptosis is tightly regulated at multiple steps to ensure that cell death occurs only when necessary, avoiding unwanted tissue damage or immune dysregulation. Regulation involves both positive (pro-apoptotic) and negative (anti-apoptotic) controls.
Key Regulatory Points and Molecules
1. Ligand Availability
-
Pro-apoptotic: FasL, TNF-α, and TRAIL promote apoptosis when expressed on immune cells or in response to stress.
-
Regulation: Controlled expression and release of these ligands prevent excessive activation.
2. Death Receptor Expression
-
Cells regulate the surface expression of:
-
Fas (CD95)
-
TNFR1
-
TRAIL-R1/DR4, TRAIL-R2/DR5
-
-
Downregulation of receptors reduces sensitivity to apoptotic signals.
3. Decoy Receptors
They lack intracellular domains and may be the products of splice isoforms or obtained by proteolytic cleavage. Decoy receptors TRAIL-R3 and TRAIL-R4 can bind TRAIL but lack intracellular death domain and therefore not able to mediate apoptosis.
-
DcR1, DcR2: Bind to TRAIL but lack functional death domains, acting as competitive inhibitors.
-
Prevent TRAIL from activating DR4/DR5, thus blocking apoptosis.
4. Inhibitors at the DISC Level
| Molecule | Function |
|---|---|
| cFLIP | Binds to FADD, blocks caspase-8 activation |
| FADD Dominant-negative variants | Prevent DISC formation |
| TRAFs (in TNFR1 signaling) | Promote survival via NF-κB pathway |
5. Regulation of Caspase Activation
-
Inhibitor of Apoptosis Proteins (IAPs):
A common feature of all IAPs is the presence of a BIR Domain (Baculovirus IAP Repeat, a ~70 amino acid domain) in one to three copies. Secondly, there is a UBA domain, which allows IAP to bind to ubiquitin. Thirdly, there is a zinc-binding domain, or a “carboxy-terminal RING Finger”. The RING domain utilizes E3 ubiquitin ligase activity and enables IAPs to catalyze ubiquination of self, caspase-3, or caspase-7 by degradation via proteasome activity. XIAP is the most potent member of IAPs. -
Activity of XIAP is blocked by binding to DIABLO (Smac) and HTRA2 (Omi) proteins released from mitochondria after proapoptotic stimuli.
Glucocorticoid-induced apoptosis
Glucocorticoids (GCs) are steroid hormones secreted from adrenal cortex and play a role as anti-inflammatory and immune-suppression. The GCs induces cell cycle arrest and apoptosis in lymphoid cells hence use in the treatment of lymphoid malignancies.




13 Comments
Sachin Sharma
May 5, 2025I just have 3 words to describe the kind of content Suraj sir & LTA provides us with :- IN-CRE-DIBLE 🏅🏆
Chanchal Choudhary
May 5, 2025Wow. Sir very simplest explain thank you
SEETA CHOUDHARY
May 5, 2025This is amazing explanation 💫💯
Shreeji Charan
May 5, 2025If you want to just study the topic – you can do it however you want to
But If you want to UNDERSTAND whole CONCEPT – join LTA “without any doubt “
Suman bhakar
May 5, 2025Amazing sir 😍💫💫🔥🔥
Lipika sahu
May 5, 2025Sir unbelievable, each and every line increadible.
Amazing explanation with detailing 🔥
Beena Meena
May 5, 2025Amazing sir 👏
Lakshay Godara
May 5, 2025Thankyou sir…
It’s best
Parul
May 5, 2025Thankyou sir. Explanation is 👌.
Mosam gurjar
May 6, 2025Thankyou sir
Amazing explanation 💫
Beena Meena
May 6, 2025Thank you sir
Seema kaur
May 6, 2025Wow sir👌🏻👌🏻…thank you
Beena Meena
May 7, 2025Thank you sir .best explanation 🙂