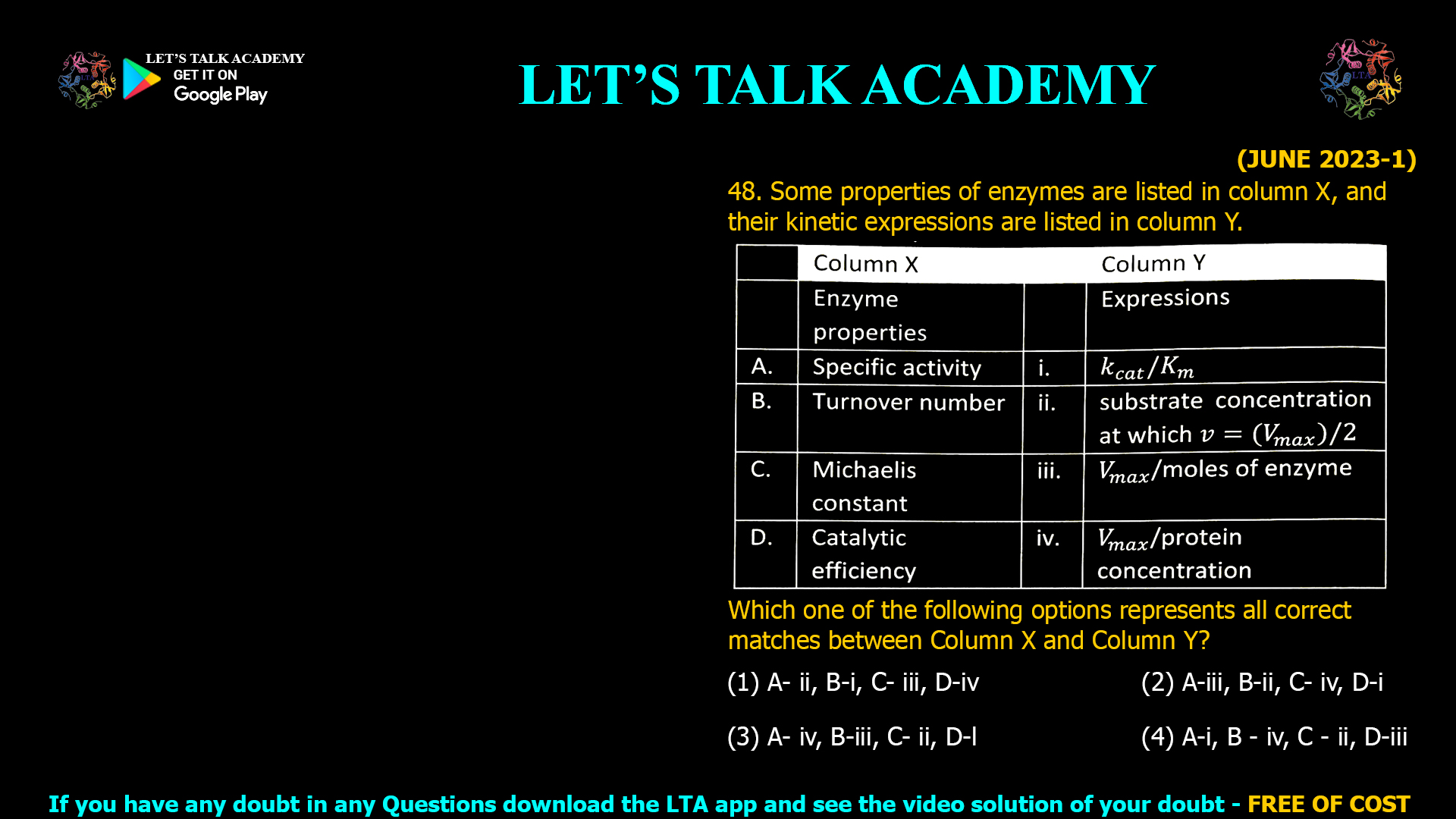
The correct answer is (3) A- iv, B- iii, C- ii, D- i. Introduction Enzyme kinetics is foundational for understanding biochemical reactions and enzyme activity. Properly identifying how various properties—such as […]
Tag: csir pyq
Tag: csir pyq
No posts found.
Common Misconceptions in Enzyme Kinetics: Understanding Allosteric Behavior, Energy Change, Inhibition, and Catalytic Efficiency
- admin
- September 12, 2025
- 35 Comments
(JUNE 2019) 47. Which one of the following statements is NOT correct? (1) Allosteric enzymes do not obey Michaelis-Menten kinetics. (2) The free-energy change provides information about the spontaneity but […]
Calculating Turnover Number and Kinetic Isotope Effect in Proton and Deuteron Transfer Enzyme Reactions
- admin
- September 12, 2025
- 12 Comments
(NOV 2020-1) 46. An enzyme catalyzed reaction comparing proton and deuteron transfer reactions yielded the following kinetic data Kcat [H], s-1 70.7 KM [H], mM 1.03 Kcat [H], s-1 10.3 […]
Analyzing Serine Protease Catalytic Efficiency and Substrate Binding: Insights from Peptide Length and Sequence
- admin
- September 12, 2025
- 17 Comments
The correct answer is (1) A and B. Introduction Enzyme kinetics provide critical insights into how substrate structure influences catalytic efficiency and binding. Serine proteases, important for protein digestion, exhibit unique […]
Analyzing Elastase Substrate Specificity Using Kinetic Data: Rate, Residue Size, and Cleavage Preferences
- admin
- September 12, 2025
- 24 Comments
The correct answer is (2) (B), (D), (E). Introduction Elastase is a proteolytic enzyme known for its specificity based on the size and nature of amino acid residues adjacent to its […]
pH Dependence of Enzyme Catalytic Efficiency: Role of Ionizable Residues in Catalysis
- admin
- September 12, 2025
- 28 Comments
(DEC 2013 GU) 43. The Kcat/Km values of an enzyme-catalyzed reaction when plotted as a function of pH yielded a bell-shaped curve with a maximum around pH 6.0. Which of […]
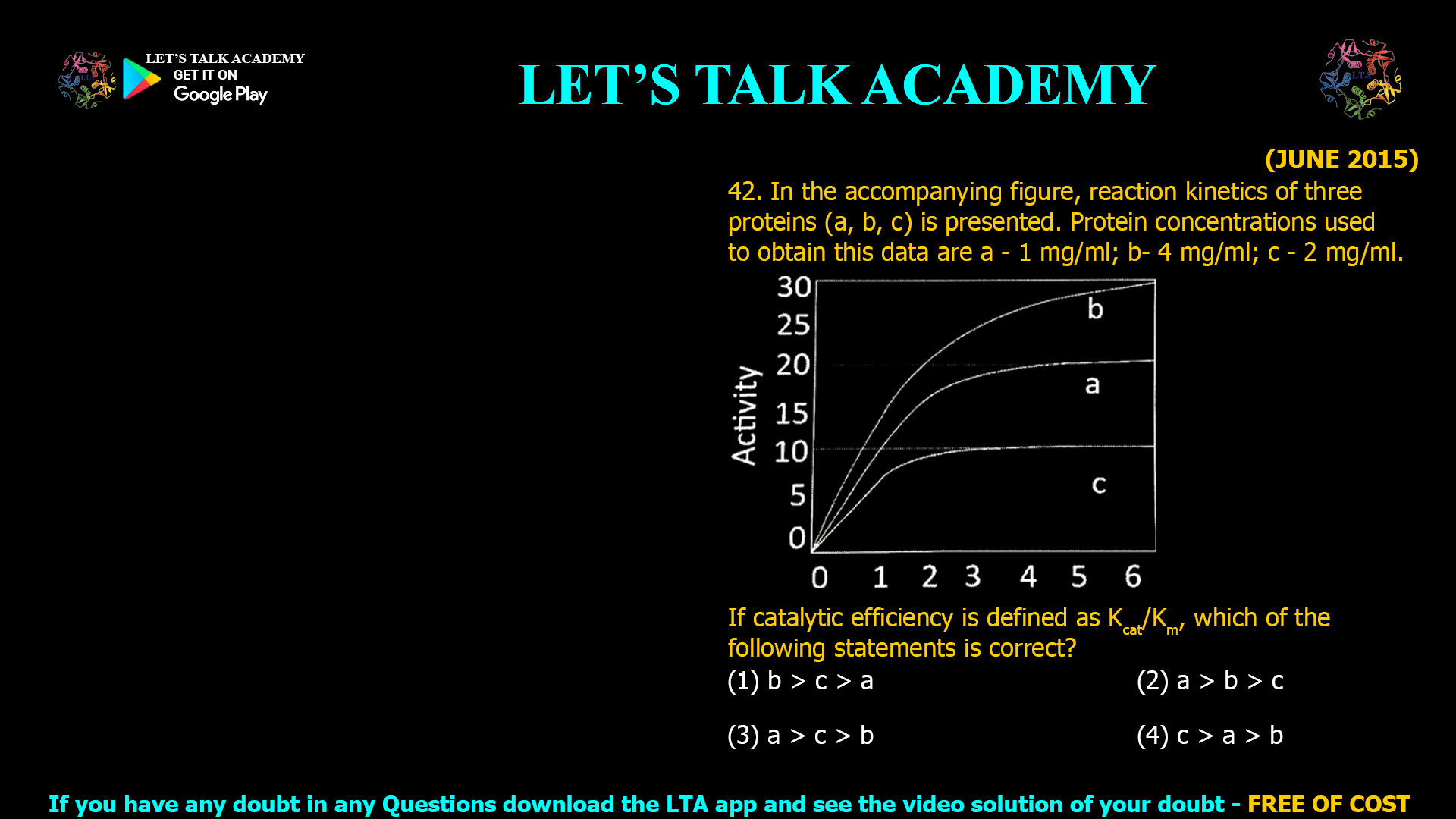
Comparing Catalytic Efficiency (kcat/Kmkcat/Km) from Enzyme Activity Curves: Analysis and Interpretation
- admin
- September 12, 2025
- 42 Comments
The correct answer is (2) a > b > c. Introduction Catalytic efficiency (kcat/Kmkcat/Km) is a key measure in enzyme kinetics, representing how rapidly and effectively an enzyme converts substrate to […]
Calculating Turnover Number (kcatkcat) for β-Amylase from Kinetic Data: A Detailed Guide
- admin
- September 12, 2025
- 12 Comments
(MODEL PAPER) 41. A solution of 1% (w/v) starch at pH 6.7 is digested by 15 µg of ß-amylase (mol wt 152,000). The rate of maltose (mol wt = 342) […]
Calculating Turnover Number (kcatkcat) for an Enzyme from Substrate and Product Data
- admin
- September 12, 2025
- 6 Comments
(JUNE 2015) 40. A 1% (w/v) solution of a sugar polymer is digested by an enzyme (20µg, MW=200,000). The rate of monomer sugar (MW=400) liberated was determined to have a […]
Calculating Turnover Number and Specific Activity of an Enzyme: Step-by-Step Guide
- admin
- September 12, 2025
- 4 Comments
(JUNE 2016) 39. The turnover number and specific activity of an enzyme (molecular weight 40,000 D) in a reaction (Vmax= 4µmol of substrate reacted/ min, enzyme amount = 2 µg) […]